Many people are coming forward with the pain and medical issues they’re experiencing with hernia mesh devices. Attorney Jason Konvicka provides a helpful FAQ below, to answer all of your questions.
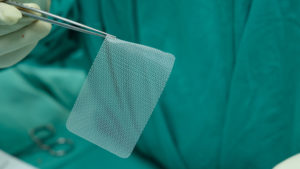
What is hernia mesh?
Hernia mesh is not a new medical device, but the use of newer designs and materials have led to an increase of adverse complications and serious injuries. Mesh was first introduced to the medical community in 1891 to repair hernia tears or bulges. A hernia occurs when an organ or fatty tissue squeezes through a weak spot in a surrounding muscle or connective tissue called fascia.
Hernias & hernia repair
Many hernias occur in the abdomen between the chest and hips, but they can also appear in the upper thigh and groin areas. Most hernias aren’t immediately life-threatening, but they don’t go away on their own. Sometimes they can require surgery to prevent dangerous complications. There are many types of hernia, mostly classified according to the physical location, with the abdominal wall being the most susceptible site. Specifically, reports show that the most frequently seen hernia is the inguinal hernia (70–75% of cases), followed by femoral (6–17%) and umbilical (3–8.5%) hernias.
Hernia repair via surgical intervention is the most common procedure performed in the world. The hernia mesh is the most common corrective measure in relieving the pain, discomfort and physical issues related to the hernia.

Is there something wrong with the hernia mesh design?
The hernia mesh is designed to hold in place the bulging intestine or internal organ. The use of hernia mesh products to surgically repair or reconstruct anatomical defects has been widely adopted. In fact, more than 80% of hernia repairs performed in United States use mesh products. The surgical mesh firmly reinforces the weakened area and provides tension-free repair that facilitates the incorporation of fibro-collagenous tissue.
However, there are many types of meshes and there is a strong controversy regarding optimum performance and the success of surgical procedures. Researchers have investigated metals, composites, polymers and biodegradable biomaterials in their quest to attain the ideal surgical mesh and implantation procedure. The sought-after characteristics are:
- Inertness
- Resistance to infection
- The ability to maintain adequate long-term tensile strength to prevent early recurrence
- Rapid incorporation into the host tissue
- Adequate flexibility to avoid fragmentation
- Non-carcinogenic response
- The capability to maintain or restore the natural respiratory movements of the abdominal wall
Hernia mesh design defects: Do I have a case?
When a mesh is removed, there will be an operative report detailing what happened with the mesh and if there were any adverse effects. Hernia mesh manufacturers have recalled more than 211,000 units of hernia mesh from 2005 to February 2019. Complications from recalled mesh devices include mesh failure, punctured organs, hernia recurrence and others. Some recalls led injured patients to file lawsuits against mesh manufacturers. The FDA has classified these recalls as Class 2, which means the damage caused by the defective medical device may not cause fatal injury.
The mesh, composed of polypropylene compound, has been known to cause the following complications:
- Adhesions
- Bowel fistulas
- Punctured organs
- Device migration
- Bowel obstruction.
Removal of the defective mesh via further surgical intervention is complex, especially since the mesh often adheres or becomes part of another organ tissue.
What hernia mesh devices are on the recall list, and being litigated?
- Atrium Medical C-QUR Mesh
- Bard Composix Kugel Hernia Patch (recall)
- Bard Inguinal Repair Perfix Plug and 3D-Max
- Bard Compsosix meshes
- Covidien Parietex Composite Parastomal Mesh, as well as Progrip
- Ethicon Proceed Surgical Mesh
- Ethicon Physiomesh Flexible Composite Mesh (complete voluntary market withdrawal)
- Ethicon Prolene System Inguinal Mesh
What do I do if I’m suffering from a hernia mesh device?
If you or a loved one have been injured by a defective hernia mesh medical device, we understand the challenges you are facing. Our product liability attorneys are available at seven convenient locations in Virginia, listed below.



